 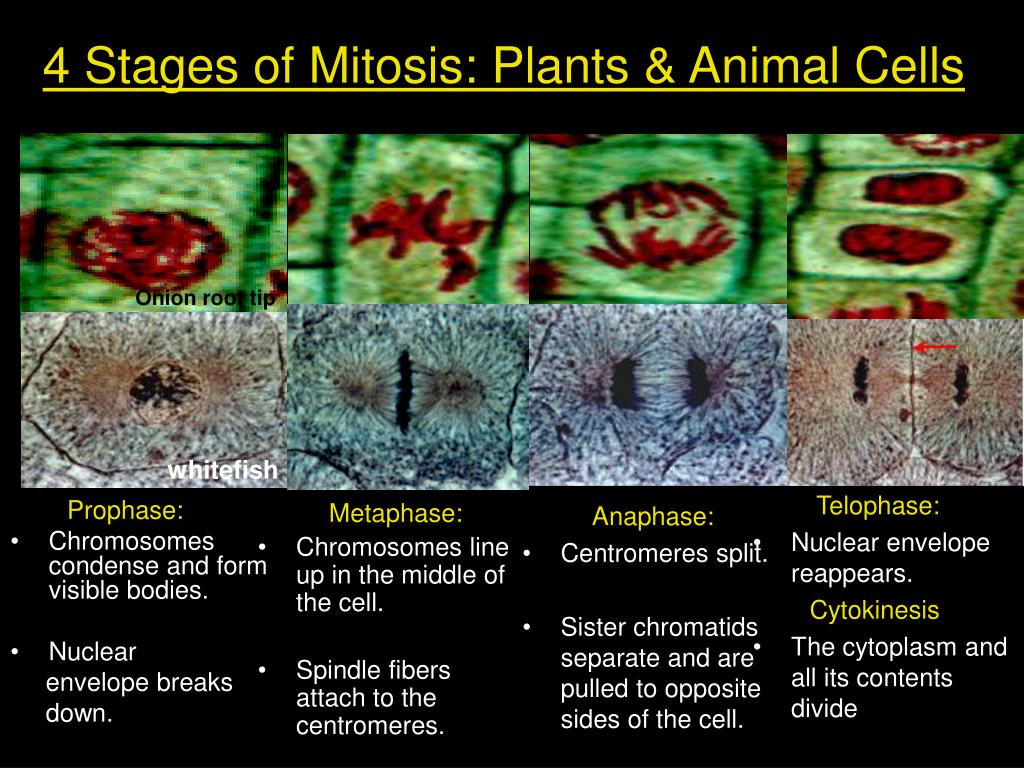
For phylogenetic analysis details see Dataset S3. See Dataset S2 for full-length 3D structures. Right: AthaBORI1 and 2 Alphafold2-predicted 3D structures, with putative phosphate-interacting residues in ball-and-sticks representation (see also C). Circles: bootstrap support (1,000× replicates, only higher than 80% support shown). Branch lengths (scaled number of substitutions per site). ( D) Unrooted maximum-likelihood phylogenetic tree of FHA domains most similar to BORI orthologs in prokaryotes and eukaryotes. Color scheme: 100% identity (black), similar physicochemical properties (gray), others (white). Stars: three arginine residues (ARG-15-32-35), which likely face the phosphorylated histone H3 tail (see ball-and-sticks representation in D). 
Secondary structure consensus of the Alphafold2 predicted 3D structures of BORI1-2 is projected below the alignment. FHA (yellow) C-terminal helix (dark blue). ( C) Multiple alignment of Borealin Related Interactor 1 and 2 found in Arabidopsis thaliana. Question mark: inability to detect orthologs. ( B) Presence–absence matrix of CPC components in model organisms that have previously been found throughout the eukaryotic tree of life ( 2, 19). Metaphase CPC localization at the centromere is dependent on a Survivin–H3T3 ph interaction and anaphase localization at the central spindle relies on interactions with microtubules and kinesins. ( A) The CPC consists of an Aurora-type kinase scaffolded by the triple helix-based trimer INCENP, Borealin, and Survivin. BOREALIN RELATED INTERACTOR ( BORI) genes in plants. Interestingly, we also detect Survivin orthologs without a defined phosphate-binding domain, likely reflecting the situation in the last eukaryotic common ancestor.įig. Using sensitive similarity searches, we find conservation of this helical domain between animals and plants and identify the missing CPC component in most eukaryotic supergroups. We find that the unifying criterion of Survivin-type proteins is a helix that facilitates complex formation with the other two scaffold components and that the addition of a phosphate-binding domain, necessary for concentration at the inner centromere, evolved in parallel in different eukaryotic groups. However, this interaction is not mediated by a BIR domain as in previously recognized Survivin orthologs but by an FHA domain, a widely conserved phosphate-binding module. Similar to Survivin, we find that the BORIs bind to phosphorylated histone H3, relevant for correct CPC association with chromatin. 
Loss of BORI function is lethal and a reduced expression of BORIs causes severe developmental defects. 
By characterizing proteins that bind to the Arabidopsis Borealin ortholog, we identified BOREALIN RELATED INTERACTOR 1 and 2 (BORI1 and BORI2) as redundant Survivin-like proteins in the context of the CPC in plants. While most CPC components are conserved across eukaryotes, orthologs of the chromatin reader Survivin have previously only been found in animals and fungi, raising the question of how its essential role is carried out in other eukaryotes. The chromosomal passenger complex (CPC) is a heterotetrameric regulator of eukaryotic cell division, consisting of an Aurora-type kinase and a scaffold built of INCENP, Borealin, and Survivin. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
